Outcome Reporting Bias in COVID-19 mRNA Vaccine Clinical Trials
Relative risk reduction and absolute risk reduction measures in the evaluation of clinical trial data are poorly understood by health professionals and the public. The absence of reported absolute risk reduction in COVID-19 vaccine clinical trials can ...

www.ncbi.nlm.nih.gov
******************************************************************************************************************************
.
.
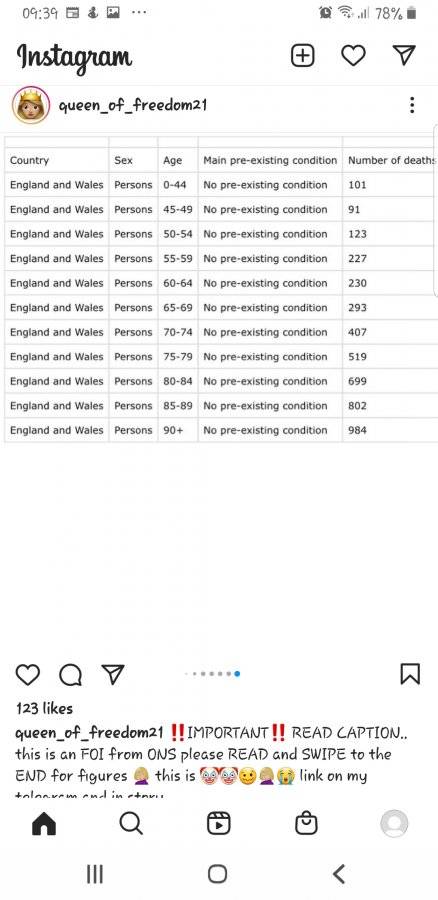
"Relative Risk Reduction from Cv19 is your risk of dying "relative" to the average person who dies with Cv19 (ie an 81 year old with 2-3 pre-existing co-morbidities). These are the risk reduction figures you see on the left - with numbers you'll be quite familiar with.
Absolute Risk Reduction from Cv19 is the average risk reduction for the average member of the population when you take the jab from the respective providers.
These data are from the same study - it's just how you decide to present them - hence the famous term, "Lies, damned lies and statistics".
Therefore, if you are an 81 year old with 2-3 pre-existing comorbidities, the recommended action seems sensible.
However, if you're not... well, you have to make that decision based on the information you're willing to find - ideally not just on the information you are given.
For more detail see the article in The Lancet:"
Approximately 96 COVID-19 vaccines are at various stages of clinical development.1 At present, we have the interim results of four studies published in scientific journals (on the Pfizer–BioNTech BNT162b2 mRNA vaccine,2 the Moderna–US National Institutes of Health [NIH] mRNA-1273 vaccine,3 the...
www.thelancet.com